News
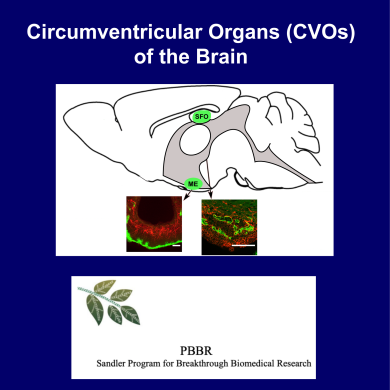 |
Mohr Receives Funds to Take on Windows of the Brain |
|
 |
Torok Appointed for Year 2 as MSK Research Fellow |
Zsofia was chosen to be one of three new Scholars on the MSK T32 training grant. She will begin looking at neuronal-non-neuronal crosstalk in the hypothalamus in regulating bone mass. |
 |
The Gut Group Strikes Again - 2025 |
Read our latest work to understand why females suffer from functional gut disorders on bioRxiv (Link). Led by two talented fellows, Drs. Archana Venkataraman and Eric Figureroa find the answers to this question. |
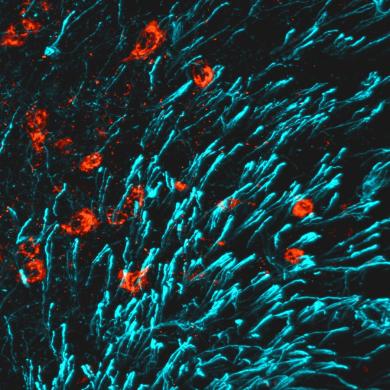 |
Our Work on a Brain-Bone Axis |
In lactating mothers, the high calcium (Ca2+) demand for milk production triggers significant bone resorption. While estrogen would normally counteract excessive bone loss and maintain sufficient bone formation during this postpartum period, this sex steroid drops precipitously after giving birth. Here, we report that brain-derived CCN3 (Cellular Communication Network factor 3) secreted from KISS1 neurons of the arcuate nucleus (ARCKISS1) fills this void and functions as a potent osteoanabolic factor to promote bone mass in lactating females. Read More in our Nature Paper. (Image of CCN3 interspersed with tanycytes in the arcuate nucleus, courtesy of Zsofia Torok). |
 |
Ingraham Receives FASEB Lifetime Award |
Please visit this link to read more on these awards. I will be accepting this award 4/2025 @APS Summit in Baltimore WHAT AN HONOR! |
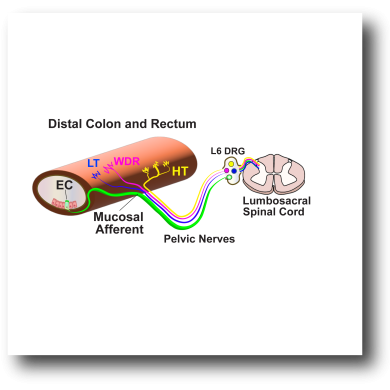 |
Our Work in Visceral Gut Pain |
The latest group effort between the Julius/Ingraham/Bayrer/Brierley Labs highlights the importance of EC cells in sex-differences in gut visceral pain and anxiety. Link to Paper and Figures |
Krause Receives R21 Funding From NIA |
Bill will be leveraging his earlier findings in the VMHvl, where he identified an estrogen-dependent activity node. Now he will ask how chronic activation of these neurons affect aging and brain health. |
|
 |
Joni Joins UC Berkeley Faculty 8/1/2023 |
Part of the lab celebrating after Joni signed his offer letter to join the faculty at UCB - he is starting his lab in the Department of Integrative Sciences in the College of Natural Resources. He hopes to be in his lab in late Fall after renovations. Check out his plans for the new lab and exciting research program aimed at understanding how the brain control lipid homeostasis (Link to website). |
 |
Ingraham receives the 2023 Lifetime Achievement in Mentoring Award |
|
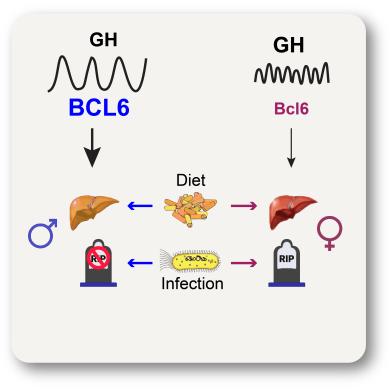 |
Joni's Paper on Trade-offs in Sex Biased Diseases (Science) |
ABSTRACT Adaptations to infectious and dietary pressures shape mammalian physiology and disease risk. How such adaptations affect sex-biased diseases remains understudied. Here, we show that sex-dependent hepatic gene programs confer a robust (~300%) survival advantage for male mice during lethal bacterial infection. The transcription factor BCL6, which masculinizes hepatic gene expression at puberty, is essential for this advantage. However, protection by BCL6 comes at a cost during conditions of dietary excess, resulting in overt fatty liver and glucose intolerance in males. Deleting hepatic BCL6 reverses these phenotypes but markedly lowers male survival during infection, thus establishing a sex-dependent tradeoff between host defense and metabolic systems. Our findings offer strong evidence that some current sex-biased diseases are rooted in ancient evolutionary trade-offs between immunity and metabolism. Accepted 8/29/22 Science |